
The violet candies represent violet photons, which have the highest energy of all visible light photons.
The red candies represent red light photons, which have the longest wavelength of all visible light.
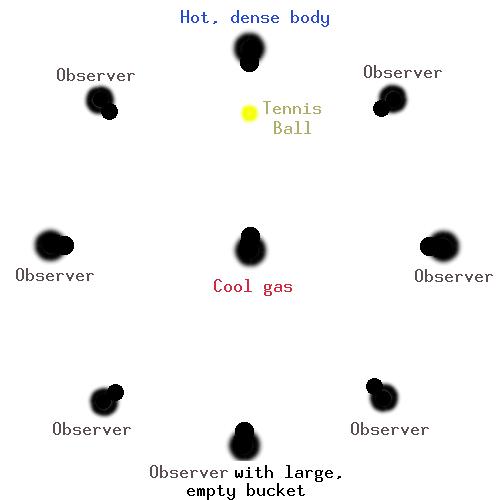
This observer will have all of the red, yellow, green, and purple (violet) candies (photons) which were tossed (emitted) by the hot, dense object. In addition, this observer will have a few blue and orange candies (photons) which were intercepted (absorbed) by the cool cloud, and then tossed (re-emitted) to them.
The cool cloud intercepts (absorbs) blue and orange candies, then tosses (re-emits) them in random directions. Since these observers are looking only at the cool cloud (they are receiving photons/candies only from the cool cloud), they will have only orange and blue candies.
In my case, both predictions were correct. In your case, one or the other or both may have been incorrect (you should specify such which was correct/incorrect in a question like this).
The observers looking only at the cool cloud see only orange and blue photons, and so see an EMISSION line spectrum.
The observer with the bucket (who is looking at the hot, dense object through the cool cloud) sees an ABSORPTION line spectrum, since they see far fewer orange and blue photons than other photons.
The hot, dense object is emitting all colours of photons, and so is emitting a CONTINUOUS spectrum.